ANXIETY
Anxiety – Large Unmet Medical Need
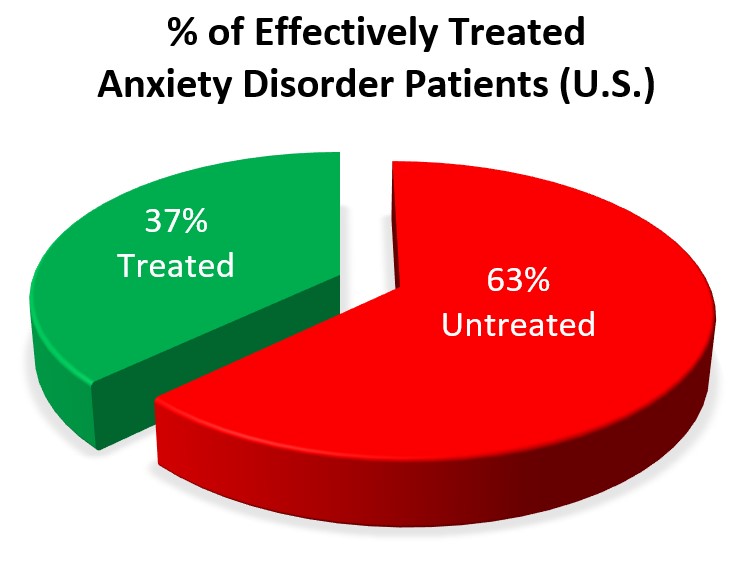
Anxiety disorders are the most common mental illness in the United States, affecting 40 million adults, or 19% of the U.S. population. Yet only 37% of anxiety disorder patients receive effective treatment. This represents one of America’s largest unmet medical needs, primarily due to the limitations of U.S. anxiety medications.
Source: Anxiety and Depression Association of America / (NIMH, 2017)
Limitations of U.S. Anxiety Medications
The top 10 U.S. anxiety drugs fall into one of two drug classes:
1) SSRIs/SNRIs – the first-line anxiety drugs, take 4-6 weeks to take effect and are not effective in over 50% of patients.
2) Benzodiazepines – which are fast and effective, but have severe side effects and are involved in 31% of U.S. fatal prescription overdoses.
This explains why over 24 million anxiety disorder patients do not receive proper treatment in the United States.
Source: Anxiety and Depression Association of America / (NIMH, 2017)
“Benzodiazepines are involved in 31% of U.S. fatal prescription overdoses.”
*Source: Centers for Disease Control and Prevention (2013 data)
Develop GRX-917 For Anxiety
GRX-917 is a deuterated (metabolically improved) analog of etifoxine, an approved anti-anxiety drug that has been sold in France and over 25 other countries for decades. Clinical studies have shown that etifoxine has equivalent speed of onset and efficacy as the world’s top two selling benzodiazepines (Xanax® and Ativan®), but with minimal to no side effects. GRX-917 has an identical mechanism of action as etifoxine.
It follows that GRX-917 is safer and clinically superior to America’s top two drug classes: SSRIs/SNRIs and benzodiazepines. And GRX-917 is not expected to be a controlled substance, since etifoxine is not a controlled substance in France or any other country in which it is sold. No clinically competitive anti-anxiety drug is on market or in late stage development, to management’s knowledge.
GABA Therapeutics is planning to develop GRX-917 for anxiety in the United States, Europe and other patented countries. GRX-917 will initially be developed for treatment of Generalized Anxiety Disorder (GAD) and then expanded into other anxiety indications. Based on the extensive, 35+ years of clinical experience with Etifoxine and its excellent efficacy and safety profile, the development of GRX-917 is considered very low risk.
