NEWS
Precision qEEG Biomarker Demonstrates Anxiolytic Efficacy in Phase 1 GRX-917 and Phase 1 Etifoxine
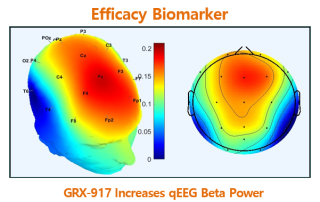
qEEG biomarkers demonstrated anxiolytic efficacy in both Phase 1 of GRX-917 and Phase 1 of etifoxine. qEEG Beta power increase Rapid-onset, sustained activity Time-dependent Exposure-dependent Increased qEEG Beta Power PD marker confirms GABAA receptor target engagement Anxiolytic efficacy biomarker For questions contact: Richard G. Farrell Chief Financial Officer Follow [...]
GABA Therapeutics Completes Single Ascending Dose (SAD) Stage of Phase 1 Study in GRX-917 in Melbourne
NEWPORT BEACH, CA – GABA Therapeutics, Inc has successfully completed the single ascending dose (SAD) stage of its Phase 1 study of GRX-917 in Melbourne, Australia. Interim data from the SAD stage showed that GRX-917 was well tolerated and favorable safety results were observed up to 500mg. Furthermore, the results [...]
GABA Therapeutics Appoints Mario David Saltarelli M.D., Ph.D. as CEO and CMO
NEWPORT BEACH, Calif., April 07, 2022 (GLOBE NEWSWIRE) -- GABA Therapeutics, Inc, (“GABA”) a clinical-stage biotechnology company developing novel therapies for anxiety, depression and other neurological disorders, and part of the atai Life Sciences (“atai”) platform, announces the appointment of Mario David Saltarelli M.D., Ph.D. as Chief Executive Officer [...]
GABA Therapeutics Completed Phase 1 Etifoxine in Melbourne
GABA Therapeutics has continued to progress the development of GRX-917 without any material impact from the COVID-19 pandemic. GABA Therapeutics recently completed its Phase 1 clinical trial of the anxiolytic etifoxine in Melbourne, Australia, on time and slightly ahead of budget. The two-stage, double-blind, placebo-controlled single and multiple dose [...]
Biotech with Anxiety Drug Lands Funding
DRUGS: $15.5M; XANAX IMPROVEMENT? Biotech with Anxiety Drug Lands Funding By Jessie Yount Orange Country Business Journal Monday, February 24, 2020 An effort to develop a new and improved version of an anti-anxiety drug—never commercialized in the U.S.—is underway in Newport Beach. Gaba Therapeutics Inc. has developed and patented a [...]
GABA Therapeutics Commences Phase I Study in Etifoxine – Melbourne Australia
Today, GABA Therapeutics commenced dosing for its Phase I clinical trial of the anxiolytic etifoxine in Melbourne, Australia – right on schedule. The Phase I, two-stage, double-blind, placebo-controlled single and multiple dose study will evaluate the pharmacokinetics, pharmacodynamics, and safety of oral etifoxine in up to 34 normal healthy [...]